LIPOPRINT®
LDL SUBFRACTIONS TEST
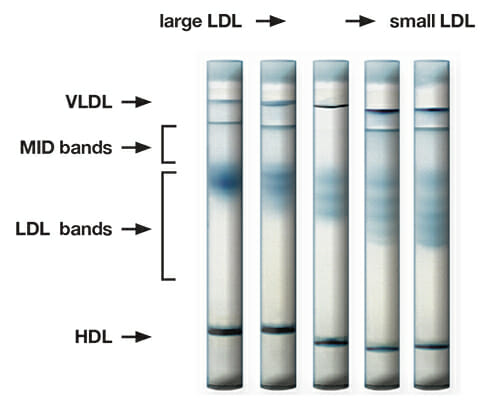
The Lipoprint system uses polyacrylamide gel electrophoresis to separate the various lipoprotein subfractions on the basis of size.
This illustrates five different patients with progressively increasing LDL subfractions distribution.
The Lipoprint Lipoprotein Subfractions Testing System from Quantimetrix is a device for in vitro diagnostic use only, intended to measure cholesterol levels in all lipoprotein fractions and LDL subfractions in fasting serum or plasma. The Lipoprint system uses polyacrylamide gel electrophoresis to separate the various lipoprotein subfractions on the basis of size. The electrophoresed gels are analyzed with Lipoware, a configured software that calculates the levels of cholesterol in each subfraction. A color coded profile is generated for ease of interpretation.
Personalized Attributes of the Lipoprint LDL Subfractions Test
To determine the clinical significance of an emerging risk factor, the ATP III requires, the following criteria: “Laboratory or clinical measurements must be widely available, well standardized, inexpensive, have accepted population-reference values, and be relatively stable biologically.”
The Lipoprint LDL subfraction test is an in vitro test device for new emerging risk factors with the following characteristics:
- The Lipoprint test is available for use in any clinical or research laboratory.
- The Lipoprint test reveals heart disease risk factors not indicated by conventional lipoprotein cholesterol measurements. It is accurate, easy to perform, and relatively inexpensive.
- The Lipoprint test was compared to established methods such as direct LDL and HDL cholesterol test methods. It was also compared to the CDC beta quantification and ultracentrifugation methods.
- Normal reference ranges for all lipoprotein fractions and LDL subfractions were derived from a normal population as defined by NCEP ATP III guidelines for desirable lipid levels.
- The test is performed on fasting serum or EDTA plasma. Samples are stable refrigerated for up to 7 days. For prolonged storage, cryogenic freezing of the samples is recommended.
